An advisory has been issued due to a labelling error of birth control pills.
According to Health Canada, the error affected one lot of Linessa 28 birth control pills on the Canadian market.

The English side of Linessa 28 boxes is incorrectly labelled with the eight-digit Drug Identification Number (DIN) for Linessa 21 (02272903) instead of the correct Linessa 28 DIN (02257238).
The products affected include Linessa 28, lot 190056, expiry 06/2021, and Linessa 28, lot 190141, expiry 06/2021.
The labelling error could result in a Linessa 21 patient inadvertently receiving Linessa 28 instead of Linessa 21 if the product is dispensed at the pharmacy only using the incorrect DIN as a reference.


Health Canada says that there is a small chance that patients who were prescribed Linessa 21 but received Linessa 28 in error may not know that the last seven green "reminder" pills in the Linessa 28 pack are inactive pills that do not contain any hormones.
If patients take a "break" week (no pills) after completing a 28-day pack, as is the practice with Linessa 21, this would result in two weeks without hormone treatment, which could increase the risk of pregnancy.
The DIN error affects the English side of Linessa 28 boxes only.
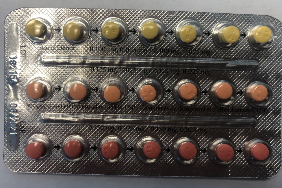
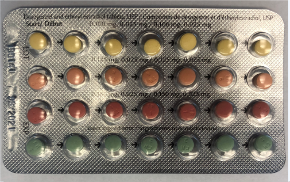
The French side of Linessa 28 boxes has the correct DIN. The product name "Linessa 28" is clearly and accurately printed on the box, the foil pouch and the blister pack.
The labelling issue does not affect the safety or quality of Linessa pills.
Patients who take Linessa 21 or Linessa 28 according to the instructions that accompany the product they received are not at an increased risk of pregnancy as a result of the error.
At Health Canada's request, Aspen Pharmacare Canada Inc. has asked that pharmacies contact patients prescribed Linessa 21 to confirm that they received the correct product.
According to the company, about 10,000 mislabelled packages from lot 190056 have been distributed nationally, starting in late May 2019. A second lot of Linessa 28 is affected by the same issue (lot 190141, expiry 06/2021) but has not yet been released on to the market.
A sticker with the correct DIN will be placed on all affected Linessa 28 boxes that have not yet been distributed, to prevent patients from receiving Linessa 28 instead of Linessa 21.